Risk Management in Aseptic Packaging

Introduction
Risk Assessment Management Aseptic Packaging plays a crucial role in ensuring product safety and sterility throughout the packaging process. It involves systematically identifying, analyzing, and mitigating potential risks such as microbial contamination, equipment malfunction, and human error. Since aseptic packaging requires both the product and packaging materials to be sterilized, even minor lapses can lead to serious issues like product recalls, regulatory non-compliance, and health risks. Implementing effective risk management strategies helps maintain quality, operational efficiency, and consumer trust.
Types of Risks in Aseptic Packaging
a) Microbiological Contamination
The biggest risk in aseptic packaging is contamination by microorganisms. If the product, the packaging material, or the equipment is not sterilized correctly, bacteria can survive and spoil the food. This not only makes the product unsafe for consumers but also results in wasted batches and financial losses..
b) Equipment Failure
Packaging lines depend on machines to sterilize, fill, and seal products. When a machine breaks down or doesn’t function properly, the process is interrupted. Faulty equipment can leave packages unsealed or unsterile, leading to both safety risks and production delays.
c) Human Error
People working on the line must follow strict hygiene and process rules. If staff are not well trained, or if they make mistakes, contamination can occur. Even simple errors—like touching a sterile surface without gloves—can compromise product safety and cause rework or waste.
d) Environmental Factors
Aseptic packaging requires a controlled environment. If clean rooms do not maintain proper air flow, temperature, or humidity, the sterile conditions are lost. Poor environmental control can damage the product’s integrity and shorten its shelf life.
e) Supply Chain Disruptions
The process also depends on a steady supply of sterile packaging materials and other inputs. Any delays in transport or issues with suppliers can stop production. Faulty or expired materials that slip through can also lead to wasted inventory.
f) Regulatory Non-Compliance
Strict food safety laws govern aseptic packaging. If a company fails to meet these requirements—whether by skipping documentation, neglecting audits, or ignoring safety standards—it risks heavy fines, product recalls, and damage to its reputation.
Risk Management Techniques
a) Hazard Analysis and Critical Control Points (HACCP)
i. Identify risks in each step of production.
ii. Set control points to monitor and prevent risks.
b) Good Manufacturing Practices (GMP)
i. Ensure hygiene, sanitation, and quality control.
ii. Regular staff training and personal hygiene monitoring.
c) Preventive Maintenance
i. Timely inspection and servicing of machinery.
ii. Reduces chance of unexpected equipment failure.
d) Environmental Monitoring
i. Use of sensors to control temperature, air quality, and humidity in sterile zones.
e) Mock Recalls and Audits
i. Test preparedness in case of contamination.
ii. Helps find weak points in the safety protocol.
f) Documentation and Traceability
i. Maintain batch-wise records of sterilization, filling, and sealing.
ii. Ensures accountability and quick corrective actions.
Preventive Measures
1. Routine calibration of sterilizers
2. Scheduled equipment maintenance
3. Validated cleaning protocols
4. Staff hygiene and training programs
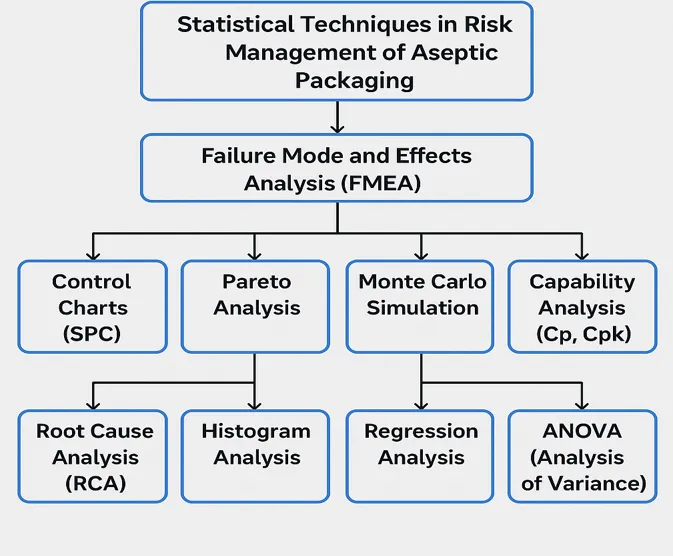
Statistical Techniques in Risk Management of Aseptic Packaging
| Statistical Technique | Purpose | Application in Aseptic Packaging |
| Failure Mode and Effects Analysis (FMEA) | Identifies possible points of failure and their impact | Used to assess potential machinery breakdown or contamination sources |
| Control Charts (SPC) | Monitors process stability over time | Tracks temperature, pressure, and sterilization consistency |
| Pareto Analysis | Highlights the most frequent or impactful issues | Used to focus on top reasons for contamination or equipment malfunction |
| Root Cause Analysis (RCA) | Finds underlying cause of problems | Helps in investigating spoilage or non-sterility cases |
| Histogram Analysis | Visualizes distribution of process data | Analyzes variability in filling volume, seal strength, or temperature control |
| Regression Analysis | Examines relationships between variables | Studies how changes in temperature or time affect microbial levels |
| Monte Carlo Simulation | Runs multiple risk scenarios to predict outcomes | Used in forecasting process risks or financial impact of potential failures |
| Capability Analysis (Cp, Cpk) | Measures how well a process meets specifications | Ensures sterile conditions and filling volumes stay within quality limits |
| ANOVA (Analysis of Variance) | Compares multiple groups or processes statistically | Evaluates differences in contamination levels between batches or equipment lines |
Example: Implementing Risk Assessment in a Milk Aseptic Packaging Plant
A dairy company is launching a new aseptic packaging line for UHT milk. The objective is to ensure the milk remains sterile and shelf-stable without refrigeration for up to 6 months.
Step-by-Step Implementation:
1. Risk Identification:
The plant team identifies the following major risks:
i. Microbial contamination during the filling process
ii. Equipment failure in the sterilization unit
iii. Temperature fluctuation in the storage area
iv. Inadequate operator training
2. Risk Analysis using FMEA (Failure Mode and Effects Analysis):
| Failure Mode | Cause | Effect | Severity | Likelihood | Detection | RPN |
| Contamination | Incomplete sterilization | Spoiled milk | 9 | 6 | 3 | 162 |
| Equipment failure | Faulty steam injector | Halt in production | 8 | 4 | 5 | 160 |
| Operator error | Lack of SOP training | Improper sealing | 7 | 7 | 4 | 196 |
Conclusion: Operator error has the highest RPN → prioritize training.
3. Statistical Monitoring (SPC Control Charts):
i. Daily temperature and pressure during sterilization are plotted using control charts.
ii. Alerts are triggered when values fall outside of ±3σ range, enabling early corrective action.
4. Preventive Action Taken:
i. Installed automatic sterilization monitors
ii. Conducted GMP (Good Manufacturing Practices) refresher courses for all operators
5. Review & Update:
i. Risks and controls reviewed monthly
ii. Updated SOPs based on observed near-misses
Outcome:
i. Zero contamination incidents in the first 6 months
ii. Improved operator efficiency by 25%
iii. Customer complaints reduced by 40%
For an in-depth understanding, please refer to our book, “Academic Research Fundamentals: Research Writing and Data Analysis”. It is available as an eBook here, or you may purchase the hardcopy here .
