How Market and Demand Trends Are Powering the Rise of Aseptic Packaging

Technical Feasibilities in Aseptic Packaging
Aseptic packaging is a highly technical process that involves packaging sterile products in a sterile environment using sterile packaging materials. The goal is to prevent microbial contamination and preserve product quality without refrigeration or chemical preservatives.
To determine its feasibility, one must assess the technical requirements across multiple dimensions: infrastructure, equipment, process control, safety, hygiene, and compliance.
Location
Location refers to the geographic placement of a manufacturing or packaging facility, strategically selected based on access to raw materials, utilities, transportation networks, skilled labor, and proximity to the target market. A well-chosen location reduces costs, ensures operational efficiency, and supports regulatory compliance.
Importance of the Location
The location where an aseptic packaging facility is set up significantly affects technical efficiency and operational cost.
1. Industrial Area Suitability: Access to zoning permissions, utilities, and waste disposal systems.
2. Transportation Connectivity: Nearby suppliers of raw materials (e.g., milk, juices, pharmaceuticals) and distribution channels.
3. Environmental Factors: Locations with minimal dust and moderate temperatures are easier to maintain in sterile conditions.
4. Skilled Workforce: Availability of trained machine operators, microbiologists, quality controllers, and maintenance technicians.
Plant Layout
A technically sound plant layout helps maintain product safety and process efficiency.
1. Sterile Zones: Packaging and filling must happen in Class 100 or ISO 5 environments.
2. Logical Flow: Movement of raw materials to production and finished goods should follow a linear or circular pattern to avoid contamination.
3. Pressure Differential Systems: Sterile areas must maintain positive air pressure compared to adjacent zones.
4. Controlled Access: Restricted entry to critical areas with airlocks, gowning zones, and sanitizing stations.
A General Idea of Aseptic Packaging
Aseptic packaging involves:
1. Product Sterilization: Typically done through UHT (Ultra High Temperature) treatment – e.g., heating milk to 135–150°C for a few seconds.
2. Packaging Material Sterilization: Achieved via hydrogen peroxide vapor, UV radiation, or high-temperature air.
3. Aseptic Environment Maintenance: Ensured using cleanrooms, HEPA filters, and strict cleaning protocols.
4. Filling and Sealing: Performed using high-speed filling machines inside sterile zones.
Machinery and Equipment
Aseptic systems rely on advanced and highly engineered equipment:
Major Components:
1. UHT Sterilizers: For product sterilization (tubular or plate heat exchangers)
2. Aseptic Fillers: Machines that can fill and seal packaging in sterile conditions
3. Packaging Sterilizers: Modules that sterilize containers before filling
4. Laminar Air Flow Units: To maintain sterility in work zones
5. CIP & SIP Systems: For automatic internal cleaning and sterilization of pipelines and machinery
Equipment Must:
1. Withstand high temperatures and cleaning chemicals
2. Be validated for sterility and performance
3. Be serviceable with minimum downtime
Good Manufacturing Practices (GMP)
GMP provides a foundation for maintaining hygiene and quality throughout the process.
Key Practices:
1. Trained Personnel: Only trained staff allowed in critical areas
2. Routine Environmental Monitoring: Checking air, surfaces, and equipment for microbial contamination
3. Validated Processes: Every step (sterilization, filling, cleaning) must be validated and documented
4. Material Handling: Use of sterile tools, containers, and gloves to avoid contamination
GMP ensures that aseptic packaging is consistently performed according to industry and safety standards.
Regulatory Compliance
Operating an aseptic packaging unit requires adherence to national and international regulatory norms. Without this, products can be rejected by both local and global markets.
Food Industry Regulations:
FSSAI, FDA, EU Regulations, ISO 22000
Pharmaceutical Regulations:
i. GMP (WHO, Schedule M)
ii. US FDA CFR 21, Part 11 (electronic records)
iii. ISO 15378 – Packaging for medicinal products
Compliance requires:
Regular third-party audits
Batch record-keeping and traceability
Validated cleaning, filling, and sealing protocols
Other Technical Considerations
Packaging Material Compatibility
The choice of packaging material is critical in aseptic processing because it must withstand sterilization methods such as heat treatment or hydrogen peroxide exposure. At the same time, it should act as a barrier against light, oxygen, and moisture, all of which can spoil sensitive products like milk or juice. Multilayer cartons, PET bottles, laminated films, and glass vials are commonly used because they balance durability, product safety, and shelf-life requirements.
Automation & Integration
Modern aseptic packaging lines rely heavily on automation to maintain speed, accuracy, and hygiene. These systems often combine filling, sealing, coding, and labeling into a single integrated process. Programmable Logic Controllers (PLCs), touch-screen HMIs, and SCADA systems make it possible to monitor performance in real time, track production efficiency, and quickly respond to alarms or deviations. This reduces human error and improves consistency.
Testing & Quality Assurance
Quality assurance is built into the aseptic process through both in-line monitoring and regular testing. Sensors continuously measure factors like fill volume, temperature, and seal strength to ensure every package meets specifications. In addition, random samples are tested for microbial safety, vacuum strength, and overall integrity. These steps confirm that sterility has been maintained and help prevent defective products from reaching the market.
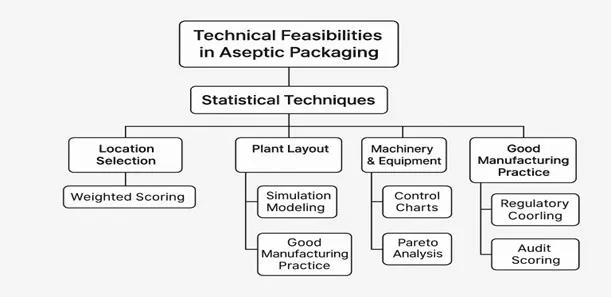
Statistical Techniques Used in Aseptic Packaging
| Component | Statistical Techniques Used | Purpose / Application |
| Location Selection | – Weighted Scoring Method- Multi-Criteria Decision Analysis (MCDA)- Correlation Analysis | To assess impact of factors like transport, workforce, and utilities on production output |
| Plant Layout | – Simulation Modeling- Flow Optimization- Time-Motion Study | To optimize space usage, material flow, and minimize contamination risk |
| Machinery & Equipment | – Statistical Process Control (SPC)- Control Charts- Capability Analysis (Cp, Cpk) | To maintain equipment performance and detect deviations in sterilization or filling |
| General Aseptic Packaging Process | – Design of Experiments (DOE)- Regression Analysis- Hypothesis Testing | To fine-tune sterilization temperatures, exposure time, and filling speeds |
| Good Manufacturing Practice | – Pareto Analysis- Frequency Distribution- Control Charts | To identify major sources of non-compliance or failures |
| Regulatory Compliance | – Audit Scoring Analysis- Compliance Trend Analysis- Statistical Sampling | To validate ongoing adherence to FDA, FSSAI, ISO standards |
| Environmental Monitoring | – Microbial Count Distribution- Standard Deviation & Variance- T-test | To assess sterility of clean rooms and packaging zones over time |
| Quality & Shelf-Life Testing | – Survival Analysis- Weibull Analysis- Descriptive Statistics | To estimate product life expectancy and packaging effectiveness under varying conditions |
Conclusion:
The steady rise of aseptic packaging is no accident—it’s a response to practical challenges faced across industries. Whether it’s keeping products safe without refrigeration or meeting consumer demand for longer-lasting goods, this method offers real advantages. By studying market trends, businesses can understand where growth is coming from and how to align their strategies. At the same time, analyzing demand helps producers focus on what matters most to end-users. When both insights come together, they provide a solid base for smart decisions and long-term success.
For deeper insights and expert market research support, visit simbi.in and discover how data-driven strategies can accelerate your business growth.
For an in-depth understanding, please refer to our book, “Academic Research Fundamentals: Research Writing and Data Analysis”. It is available as an eBook here, or you may purchase the hardcopy here .
